Understanding Diabetic Foot Ulcers: Why Proper Wound Care Is Critical
Diabetic foot ulcers are one of the most serious and challenging complications associated with diabetes mellitus. Affecting approximately 15% of all people living with diabetes at some point in their lifetime, these wounds can quickly escalate from minor skin breaks to deep, infected lesions that threaten limb — and even life — if not properly managed. In the United States alone, diabetic foot ulcers are responsible for more than 80,000 lower-extremity amputations each year, making them a leading cause of disability among diabetic patients.
The stakes couldn’t be higher. Yet with the right wound care strategies, regular monitoring, and a proactive approach to prevention, the majority of diabetic foot ulcers can be healed successfully. This comprehensive guide walks you through everything you need to know about wound care for diabetic foot ulcers — from understanding why they develop to the most advanced treatment options available today.
Whether you are a patient living with diabetes, a caregiver, or a healthcare professional seeking a thorough refresher, this article will equip you with the knowledge and tools necessary to approach diabetic foot ulcer management with confidence and competence.
What Are Diabetic Foot Ulcers?
A diabetic foot ulcer (DFU) is an open sore or wound that occurs on the feet of people with diabetes. These ulcers most commonly develop on the bottom of the foot — particularly beneath the big toe and the balls of the feet — but they can also form on the sides, top, and heel of the foot. They vary in depth, ranging from superficial breaks in the skin to deep wounds that reach muscle, tendon, and bone.
Diabetic foot ulcers are classified using several grading systems. The most widely used is the Wagner Ulcer Classification System, which grades wounds from 0 to 5:
- Grade 0: No open lesions; skin intact with possible pre-ulcerative changes
- Grade 1: Superficial ulcer involving the full skin thickness but not underlying tissue
- Grade 2: Deep ulcer penetrating to ligaments, tendons, or joint capsule
- Grade 3: Deep ulcer with abscess, osteomyelitis, or joint sepsis
- Grade 4: Localized gangrene (toes, forefoot, or heel)
- Grade 5: Extensive gangrene involving the entire foot
Early identification and prompt treatment at Grades 1 and 2 are crucial for preventing progression to more severe, limb-threatening stages.
Why Do Diabetic Foot Ulcers Develop?
Understanding the root causes of diabetic foot ulcers is fundamental to both treating them effectively and preventing their recurrence. Several interconnected physiological mechanisms put people with diabetes at significantly elevated risk.
Peripheral Neuropathy
High blood sugar levels over time damage the peripheral nerves, resulting in a condition called peripheral neuropathy. This nerve damage causes numbness, tingling, and loss of sensation in the feet. When a person can no longer feel pain, pressure, or heat normally, small injuries — a pebble in the shoe, a blister from ill-fitting footwear, or a minor cut — go unnoticed and untreated, eventually progressing into full-blown ulcers.
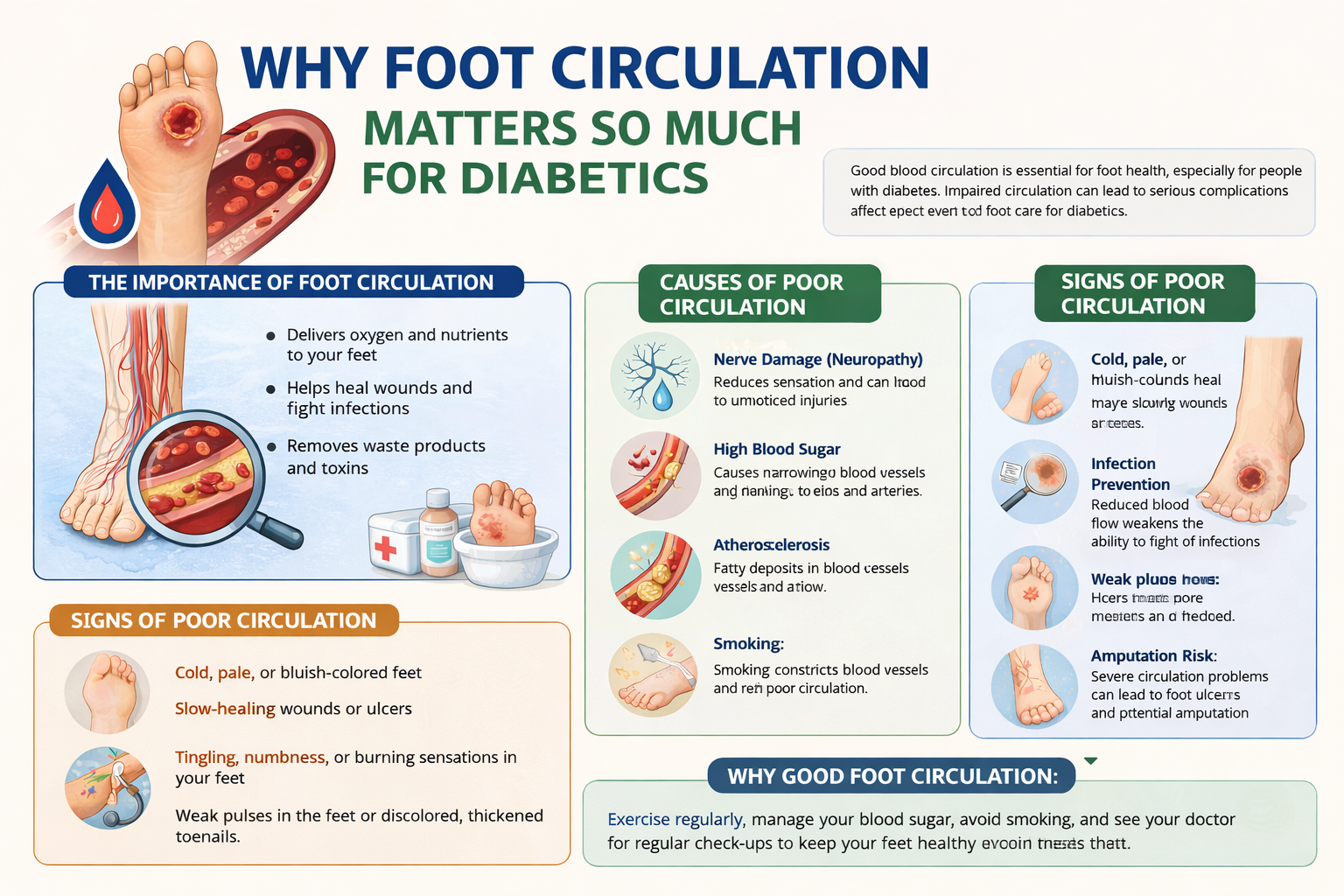
Peripheral Artery Disease (PAD)
Diabetes significantly increases the risk of peripheral artery disease, a condition in which narrowed arteries reduce blood flow to the legs and feet. Adequate blood circulation is essential for delivering oxygen, nutrients, and immune cells to healing wounds. When blood flow is compromised, the body’s ability to repair damaged tissue is severely diminished, and even minor wounds struggle to heal.
Impaired Immune Function
Elevated blood glucose levels impair the function of white blood cells, which are the body’s primary defenders against infection. This means that when a wound does develop, the diabetic patient’s immune system is less equipped to fight off bacteria, significantly increasing the risk of wound infection and complications.
Structural Foot Deformities
Diabetes-related nerve and tissue damage can alter the shape of the foot over time, creating deformities such as Charcot foot, hammertoes, and bunions. These structural changes create abnormal pressure points, friction areas, and bony prominences that are highly susceptible to breakdown and ulceration.
Skin Changes
Dry, cracked skin is common in diabetic patients due to autonomic neuropathy affecting the sweat glands. Dry, thickened skin — particularly calluses — can break down under pressure and become entry points for bacteria and ulcer formation.
Recognizing the Signs of a Diabetic Foot Ulcer
Because neuropathy often reduces or eliminates sensation, many diabetic patients do not realize they have a foot ulcer until it has already progressed significantly. This is why daily foot inspection is so critical. Common signs and symptoms to watch for include:
- Visible open sore or wound on the foot, often surrounded by thick callus tissue
- Drainage or discharge from the wound — may be clear, yellow, or foul-smelling
- Redness, swelling, or warmth around the wound site
- Skin discoloration — darkening or blackening of tissue may indicate tissue death (gangrene)
- Foul odor emanating from the wound or foot
- Fever or chills — signs of systemic infection requiring immediate medical attention
- Slow-healing or non-healing wounds that persist for more than two weeks
If you notice any of these signs, seek medical evaluation immediately. Early intervention is the single most important factor in preventing amputation.
The Wound Care Process: Step-by-Step Management
Proper wound care for diabetic foot ulcers is a multi-faceted process that involves cleaning, debridement, dressing selection, infection control, and offloading. Below is a detailed breakdown of each component of an effective wound care regimen.
Step 1: Medical Assessment and Wound Evaluation
Before any wound care begins, a thorough medical assessment is essential. A healthcare provider — ideally a wound care specialist, podiatrist, or diabetologist — should evaluate the wound to determine its grade, depth, size, and the presence of infection. Key diagnostic steps include:
- Wound measurement: Documenting the length, width, and depth of the ulcer using a sterile probe
- Vascular assessment: Evaluating blood flow through ankle-brachial index (ABI) testing, Doppler ultrasound, or other vascular studies
- Neurological testing: Assessing sensation with a 10-gram monofilament and vibration testing
- Laboratory tests: Blood glucose levels, HbA1c, complete blood count, and inflammatory markers
- Imaging: X-rays or MRI to detect bone infection (osteomyelitis) or gas in the tissue
- Wound culture: Identifying the bacteria present in infected wounds to guide antibiotic therapy
Step 2: Debridement — Removing Dead and Infected Tissue
Debridement is the process of removing dead, damaged, or infected tissue from the wound to promote healing. It is one of the most critical steps in diabetic foot ulcer management, as necrotic tissue prevents healthy tissue from growing and creates a breeding ground for bacteria.
There are several types of debridement used in clinical practice:
- Sharp/Surgical Debridement: The use of scalpel, scissors, or curette to physically remove necrotic tissue. This is the gold standard for most diabetic foot ulcers and provides immediate results.
- Enzymatic Debridement: Topical enzyme preparations (such as collagenase) applied to the wound to break down and dissolve dead tissue. Useful for wounds where surgical debridement is not feasible.
- Autolytic Debridement: Using moisture-retentive dressings to allow the body’s own enzymes to break down devitalized tissue. Slow but suitable for non-infected wounds with minimal necrosis.
- Mechanical Debridement: Wet-to-dry dressings or hydrotherapy to physically remove devitalized tissue. Less selective and can also damage healthy tissue.
- Biological (Maggot) Therapy: The use of sterile maggots to selectively consume necrotic tissue. Effective in chronic wounds, particularly those with significant bioburden or methicillin-resistant Staphylococcus aureus (MRSA).
Regular, serial debridement is often necessary, as diabetic foot ulcers tend to accumulate necrotic tissue and callus rapidly. In many wound care programs, debridement is performed at every clinic visit.
Step 3: Wound Cleansing
Proper wound cleansing removes debris, bacteria, and exudate from the wound bed without damaging fragile new tissue. Key principles include:
- Use normal saline (0.9% sodium chloride) or commercially available wound irrigation solutions for gentle cleansing
- Apply cleansing solution with gentle pressure — approximately 8 PSI — using a 35 mL syringe and 18-gauge needle or dedicated wound irrigation device
- Avoid antiseptics such as hydrogen peroxide, povidone-iodine, or Dakin’s solution on healing wounds, as these can be cytotoxic and damage new granulation tissue
- Clean the periwound skin (area surrounding the wound) to remove biofilm and prevent bacterial spread
Step 4: Infection Control and Antimicrobial Management
Infection is one of the most serious complications of diabetic foot ulcers and can rapidly lead to osteomyelitis, sepsis, and amputation. Effective infection management involves both local and systemic approaches.
Signs of wound infection include:
- Increased redness, warmth, and swelling
- Purulent (pus-containing) drainage
- Foul odor
- Wound deterioration despite appropriate care
- Elevated white blood cell count and inflammatory markers
Local antimicrobial strategies include:
- Silver-containing dressings: Dressings impregnated with ionic silver provide broad-spectrum antimicrobial activity against bacteria including MRSA
- Iodine-based dressings: Cadexomer iodine dressings slowly release iodine to kill bacteria while absorbing exudate
- Honey dressings: Medical-grade Manuka honey has potent antimicrobial and anti-inflammatory properties
- Biofilm management: Many chronic DFUs harbor biofilms — structured communities of bacteria encased in a protective matrix. Specialized debridement and antimicrobial agents are required to disrupt biofilm
Systemic antibiotics are indicated when there is evidence of spreading infection, cellulitis, systemic signs of infection, or bone involvement. Antibiotic selection should be guided by wound culture and sensitivity results. Common organisms include Staphylococcus aureus, Streptococcus species, Enterococcus, and gram-negative bacteria. Osteomyelitis typically requires prolonged antibiotic therapy (4–6 weeks or longer) and may necessitate surgical bone debridement.
Step 5: Wound Dressing Selection
Choosing the appropriate wound dressing is a critical decision that significantly impacts healing outcomes. The ideal dressing should maintain a moist wound environment, manage exudate, protect from further contamination, and be comfortable for the patient. There is no single “best” dressing for all diabetic foot ulcers — selection depends on wound characteristics, exudate level, presence of infection, and wound depth.
Common wound dressing categories and their uses:
- Hydrocolloids: Semi-permeable dressings that maintain moisture and promote autolytic debridement. Best for shallow, minimally exuding wounds.
- Hydrogels: Water-based gels that add moisture to dry wounds and facilitate autolytic debridement. Ideal for necrotic or dry wounds.
- Alginates: Seaweed-derived dressings with high absorbency. Excellent for heavily exuding wounds.
- Foam Dressings: Highly absorbent, cushioning dressings for moderate-to-heavy exudate. Often used as secondary dressings.
- Silver Dressings: Antimicrobial dressings for infected or at-risk wounds. Available in foam, alginate, and other formats.
- Collagen Dressings: Provide structural scaffold for tissue regeneration. Useful for chronic, non-healing wounds.
- Composite Dressings: Multi-layer dressings combining various properties for complex wounds.
Dressing changes should follow the manufacturer’s guidance and clinical judgment, typically ranging from daily to every 3–7 days depending on the product and wound status. Frequent unnecessary dressing changes can disrupt the healing wound bed and cause patient discomfort.
Step 6: Offloading — Relieving Pressure from the Wound
Offloading — redistributing or completely relieving pressure from the ulcer site — is arguably the most important, yet most frequently underutilized, component of diabetic foot ulcer treatment. Without adequate pressure relief, even the best wound dressings and medications will fail, because the repetitive mechanical stress of walking continues to traumatize the wound with every step.
Offloading options include:
- Total Contact Cast (TCC): Considered the gold standard for offloading plantar diabetic foot ulcers. A custom-fitted cast that distributes weight evenly across the foot and lower leg, virtually eliminating pressure at the ulcer site. It is non-removable (or minimally removable), which promotes compliance.
- Removable Cast Walker (RCW): A removable boot that provides good offloading. Less effective than TCC due to patient non-compliance (removing the device when at home or at night).
- Instant Total Contact Cast (iTCC): An RCW that has been rendered irremovable (by wrapping with a layer of fiberglass cast material). Matches TCC in effectiveness.
- Half Shoes and Healing Sandals: Custom footwear devices that offload specific areas of the foot. Suitable for heel ulcers or when casting is not possible.
- Custom Orthotics and Therapeutic Footwear: Custom-molded insoles and shoes designed to reduce plantar pressure at high-risk sites. Primarily used for prevention after healing rather than active treatment.
- Bed Rest and Wheelchair Use: Complete non-weight-bearing for severe cases,
Categories: